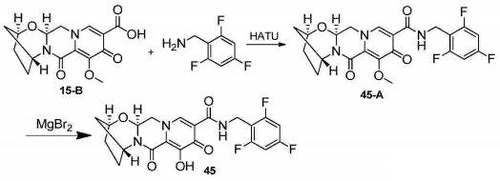
Bictegravir, is a novel, potent inhibitor of HIV-1 integrase with an IC50 of 7.5 nM.
Bictegravir is used in trials studying the treatment of HIV-1 and HIV-2 infection.
It has been approved for HIV-1 monotherapy combined with 2 other antiretrovirals in a single tablet.
Bictegravir is indicated in the management of HIV-1 infection in patients not previously treated with antiretroviral therapy.
Additionally, Bictegravir is indicated in the management of HIV-1 infection in patients who are virologically suppressed (HIV-1 RNA <50 c/mL) on a regular antiretroviral regimen for a minimum of three months without a history of failure in treatment and no known factors associated with the resistance to the individual components of the medication. It is used in combination with tenofovir and emtricitabine.
Bictegravir is an HIV-1 integrase strand transfer inhibitor (INSTI).
Bictegravir (BIC) inhibits HIV-1 virus replication into the human genome.
Bictegravir (BIC) inhibits strand transfer of viral DNA into the host genome and thereby prevents HIV-1 replication.